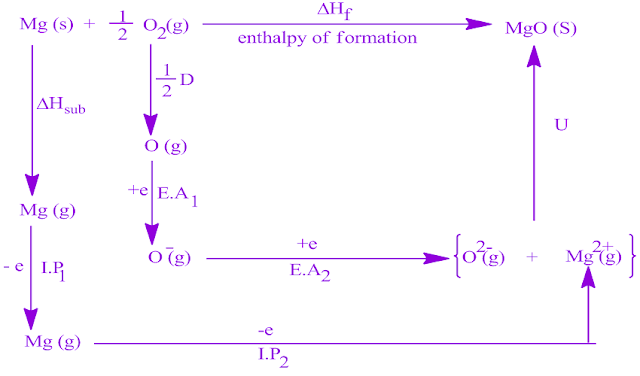
The energy required to remove one electron from the valance shell of an atom in its gaseous state is called its first ionization energy. Atoms that tend to lose electrons thus get a positive charge. Born-Haber cycle is an application of Hess’s cycle which states that the enthalpy change in a chemical change is independent of the path by which the chemical change is taking place.Ītom can lose or acquire electrons to form ions and they do so to complete their valance shell electronic configuration and complete their octet in their outermost shell.This lattice enthalpy is measured by Born-Haber Cycle.  
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
